A Practical Look at Sample Size Re-Estimation of the Promising Zone Design

A Practical Look at Sample Size Re-Estimation of the Promising Zone Design
The randomized controlled clinical trial (RCT) with a fixed design has become a gold standard for evaluating drug safety and efficacy since its debut. However, because of several drawbacks of RCTs, they have failed to meet the need for flexibility and adaptivity in modern drug development. For instance, sample sizes cannot be adjusted for a lower-than-expected treatment effect size once the study has started. Adaptive study designs that allow early access to drug efficacy and adjustment to the study are desired for improving trial efficiency.
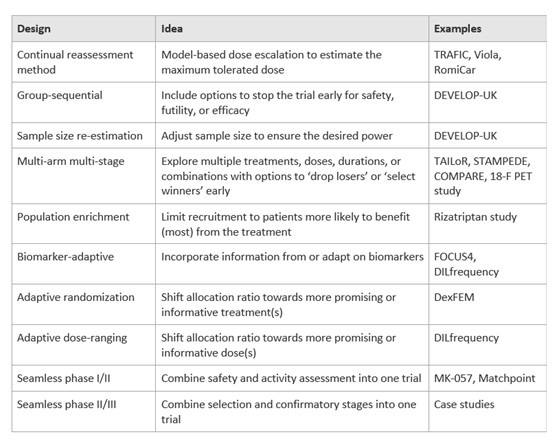
To date, many adaptive designs have been developed and implemented in actual trials (Table 1), e.g., group sequential design (GSD) that allows pre-specified interim analyses and a decision is made at each interim to stop the trial early for either futility or efficacy. The phrase “interim analysis” refers to an evaluation of current data from an ongoing trial that addresses the primary research question. Interim analyses are leveraged in RCTs to introduce flexibility and have the potential to change the way the study is conducted, i.e., adaptive design.
Table 1 overview of adaptive designs with examples of trials employed these methods (Pallmann et al., 2018)
Despite many of the promising features, the uptake of adaptive designs in clinical trials remains low, mainly due to statistical and operational challenges. For instance, the more interim looks, the more penalty it takes at the final analysis in a GSD, which will likely result in a larger sample size than a fixed design.
An alternative adaptive design to GSD that allows adjusting the sample size during the study without paying any penalty at the final analysis is sample size re-estimation with the promising zone design (SSR) introduced by Mehta and Pocock (2011). This design allows re-evaluation of the sample size based on the evidence accumulated at the interim analysis to determine whether an increase in sample size is needed to reach the desired statistical power. The additional sample size is invested only when the interim results are promising. As such, it greatly increases the chances of eventual success and reduces the risk of an expensive failed study. For more details about the benefit of the SSR design compared to a fixed design or a GSD, please refer to Mehta and Pocock’s paper.
Application
Trial operating characteristics elucidate how a design performs under various scenarios (assumptions), e.g., the power to detect a clinically significant treatment effect, and are often required in the protocol by regulatory authorities for complex adaptive designs. Trial operating characteristics are usually studied by clinical trial simulation, which is an in silico virtual experiment without the actual running of the trial. With better understanding of the trial operating characteristics, informed decisions can be made by the sponsor for improved trial efficiency. For more details about clinical trial simulations, please refer to this article. We will leverage the clinical trial simulation to understand the trial operating characteristics of a SSR design in the following example, i.e., to understand the relationship among treatment effect, maximum affordable sample size, the timing of the interim analysis, false positive (type 1 error) rate and power (1 – type 2 error rate).
In this practical guide, we specifically discuss unblinded SSR with the promising zone design. This design, among many other adaptive designs, e.g., GSD, mainly relies on conditional power. Conditional power is the probability of rejecting the null hypothesis conditioned on the evidence accumulated at the interim analysis, i.e., the interim estimate of the drug effect size. Once calculated, the conditional power is used to define the boundary of the promising zone. When the interim result is showing positive signal, or in other words, “promising”, we boost the sample size to reach the desired statistical power. If the interim conditional power is either too low or too high (i.e., unfavorable – positive result will be next to impossible, or favorable – highly likely to yield positive result, respectively), the study proceeds with the planned sample size.
Three key parameters are to be defined for SSR: 1. timing of the interim analysis, e.g. when 50% of subjects have reported the primary endpoint data, 2. Maximum affordable sample size, e.g. 1.5 times of the initial sample size, 3. Initial sample size calculated (Figure 1).
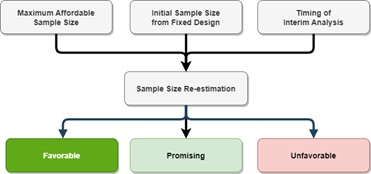
Figure 1. Flow diagram of key parameters for re-estimating the sample size at the interim analysis
We will walk through a case study to give you a better idea how SSR is applied. Assuming a two-arm trial comparing the treatment effect of an anti-viral drug vs. the placebo is to be conducted. The typical two-sided type 1 error rate of 0.05 and power of 90% are assumed. Assuming that the treatment effect is 0.07, from the literature, then a total sample size of 264 subjects is needed for a fixed design. However, the study sponsor admits that an effect of 0.07 is uncertain and can be over-optimistic and that a more realistic assumption can be anywhere between 0.04 (minimally clinically significant difference) and 0.07. We therefore introduce the SSR design to meet the need for dynamic adjustment of the sample size for uncertain treatment effects, i.e., if the treatment effect is less than expected, we will still have the chance to increase the sample size to ensure the success of the trial (the statistical power). The sponsor places a limit on funding up to 1.75 x the sample size of 264, i.e., 462. This is the maximum affordable sample size. With the last assumption that the sample size will be re-estimated when 50% of the subjects report the primary outcome, i.e., 132 subjects, we are able to perform the interim look at the data and decide whether we need to increase the sample size.
Based on these assumptions, show there are 31.4% probability that the conditional power will be in the “promising zone”, thus, resulting in a sample size increase. The % increase in sample size given the conditional power at the interim analysis is plotted in figure 2: if the conditional power of the actual trial data falls within this “promising” zone, the sample size will be increased to a maximum of 1.75 times the originally planned sample size to maintain the desired power of 90%.
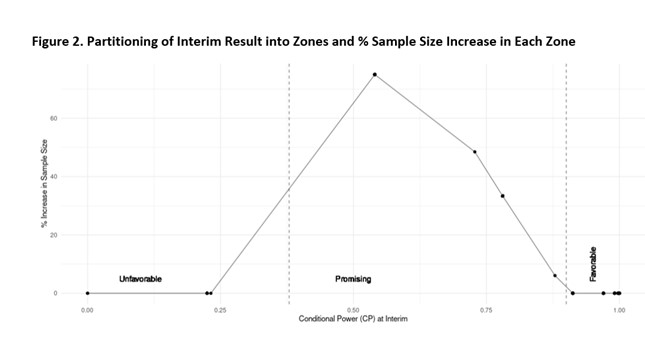
We can further alter the effect sizes and/or timing of the interim analysis to calculate the under these alternative assumptions. The optimal design can then be chosen among these alternatives.
To fully leverage the benefits of SSR promising zone design, or for more insights about SSR or other adaptive designs, please do not hesitate to contact us.
Reference
Pallmann, P., Bedding, A.W., Choodari-Oskooei, B. et al. Adaptive designs in clinical trials: why use them, and how to run and report them. BMC Med 16, 29 (2018). https://doi.org/10.1186/s12916-018-1017-7
Mehta, C.R. and Pocock, S.J. (2011), Adaptive increase in sample size when interim results are promising: A practical guide with examples. Statist. Med., 30: 3267-3284. https://doi.org/10.1002/sim.4102
CONTACT
ALIMENTIV STATISTICS
Providing 40 years of service, Alimentiv Statistics is a niche Contract Research Organization focused on Biostatistics, Statistical Analysis, Clinical Trial Design and Data Management. We invite you to see how we exceed our clients’ expectations.
